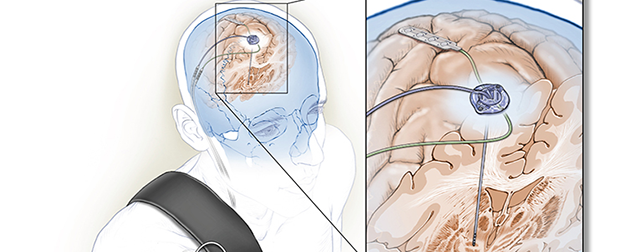
Scientists at the University of California, San Francisco (UCSF) have discovered a new way to use Deep brain stimulation (DBS) for Parkinson’s disease management that is more effective than the conventional DBS method.
In Parkinson’s disease Deep Brain Simulation has been a standard therapy that works fairly well in most but not all people. It’s been around about 20 years and it hasn’t changed much in twenty years.
Scientists are scrambling to find ways to ease the symptoms of Parkinson’s or even cure it. Wearable Technologies recently reported on a collaboration between Michael J. Fox Foundation for Parkinson’s Research (MJFF) and Verily Life Sciences LLC to monitor Parkinson’s patients.
In DBS, scientists surgically implant an electrode into the brain to manage Parkinson’s disease symptoms. Traditional approach of DBS provides constant stimulation to a part of the brain called the basal ganglia to help ease the symptoms of Parkinson’s. However, this method can lead to unwanted side effects, requiring reprogramming by a trained clinician.
So the scientists were interested in improving the way the therapy works.
Read more Michael J. Fox Foundation’s Collaboration with Verily Aims to Deepen Understanding of Parkinson’s
“Right now DBS is a pretty crude therapy; it’s a stimulator that’s always on not responding to changing needs of the brain, which do change during the day or as time goes on in patients,” says lead researcher Dr. Philip Starr of UCSF.
The new method described in this study is adaptive, so that the stimulation delivered is responsive in real time to signals received from the patient’s brain.
Researchers at University of California, San Francisco used an implanted Medtronic deep brain stimulator that can sense brain activity, process it, and quickly adjust the stimulation delivered.

“We are using an investigational new device that’s made by Medtronic that delivers standard stimulation therapy, but also has an extra circuit that can sense and store brain activity,” says Dr. Starr.
The researchers managed to decrease the power consumption of the implant by nearly half while maintaining the effectiveness of the therapy, which means the implant can last longer. Moreover, it points to the ability to actually enhance the therapy in a meaningful way for the patient.
Read more Kessler Foundation and Virtualwear Team Up to Improve Recovery Outcomes in Stroke Patients
“We first are detecting specific signals in patients who are getting DBS therapy – signals that relate to their symptoms, and then we’re using those symptoms automatically adjust the level of stimulation. So we’re prototyping feedback controlled brain simulation,” Dr. Starr said.
Many patients with Parkinson’s disease who would be helped by DBS are hard to treat because too much stimulation can trigger dyskinesia. Thus, finding the precise level of stimulation is like trying to hit a target that’s constantly moving. An adaptive system like the one in this new study could give us an efficacious alternative and may also curb negative effects of conventional method of deep brain stimulation. However, substantial testing remains to be done.
“We are now planning larger, longer-term trials to determine how effective this system is in managing the symptoms of patients with Parkinson’s disease,” Dr. Starr concluded.
This study was supported by the NIH’s BRAIN Initiative, the NINDS, the UC President’s Postdoctoral Fellowship, and a NDSEG graduate fellowship.