Hydrocephalus, a condition in which there is an accumulation of cerebrospinal fluid (CSF) within the brain, can be treated with a shunt system, and this treatment often includes complications.
Researchers at Northwestern University have developed a new wearable shunt monitor that may revolutionize the way people with hydrocephalus are cared for.
Read more Sweat-Based Wearable Sensor May Soon Replace Blood Tests
The current standard of care involves the surgical implantation of a straw-like catheter known as a ‘shunt,’ which drains the excess fluid out of the brain and into another part of the body. However, these shunts have a nearly 100% failure rate over ten years, and it’s very difficult to diagnose shunt failure.
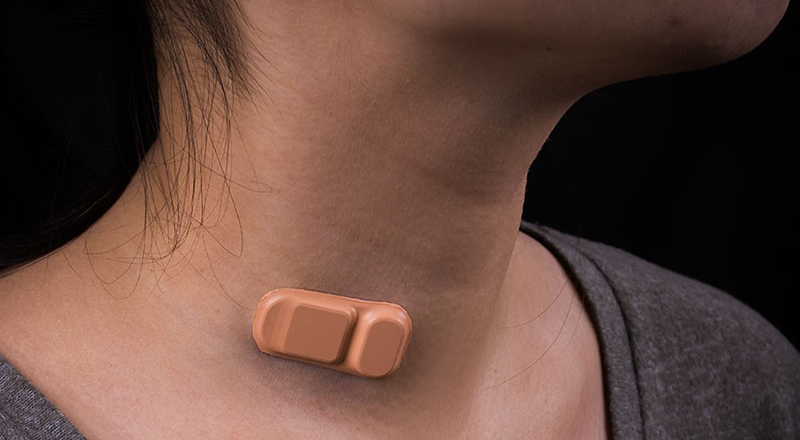
A new wireless, Band-Aid-like sensor developed by Northwestern researchers could revolutionize the way patients manage hydrocephalus and potentially save the U.S. health care system millions of dollars, reports Northwestern.
The device was successfully tested on 5 adult patients with hydrocephalus in a Northwestern Medicine clinical study.
Within five minutes of placement of the device, the sensors allowed patients to determine if fluid was flowing through their shunt. This non-invasive, flexible sensor uses measurements of temperature and heat transfer to tell if and how much fluid is flowing through.
“We envision you could do this while you’re sitting in the waiting room waiting to see the doctor,” said co-lead author Siddharth Krishnan, a fifth-year Ph.D. student in the Rogers Research Group. “A nurse could come and place it on you and five minutes later, you have a measurement.”
A tiny rechargeable battery is built into the sensor. The Bluetooth enabled device is able to communicate with a smartphone and transmit the readings via an Android app. The sensor advances concepts in skin-like “epidermal electronics,” which the Rogers Research Group has been working on for nearly a decade.
The sensor uses small amounts of thermal power to minimally increase the temperature of the skin, a procedure known as thermal transport measurement.
The sensor measures a characteristic heat signature if the shunt is working and the excess cerebral spinal fluid is draining properly. On the other hand, if the shunt malfunctions and there’s no flow, the sensor is able to quickly indicate that via heat flow measurements.
Read more This Wearable Can Measure Your Stress Through Stress Hormones in Your Sweat
There will be a larger pediatric clinical trial at Ann & Robert H. Lurie Children’s Hospital of Chicago. The researchers are also working on outsourced production on the scale of a few hundred sensors to support this study and further develop the technology.
The findings of the study were published in Science Translational Medicine.